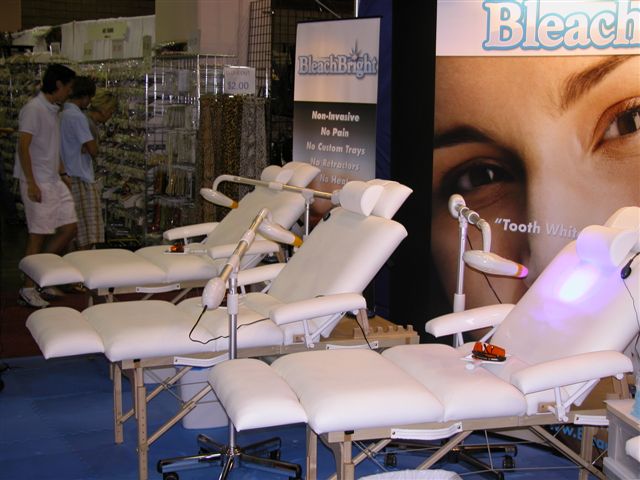
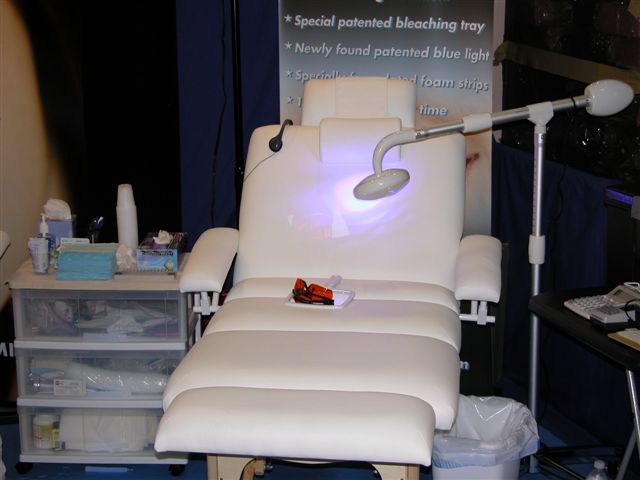
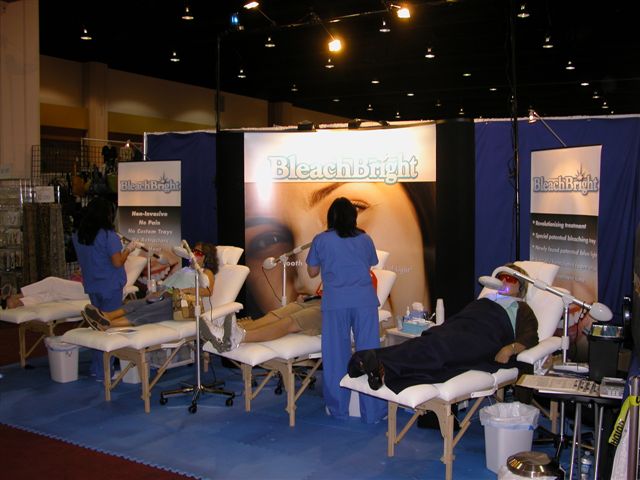
|
Federal Guidelines:
FDA:
RULES,
GUIDANCE, REGULATIONS AND CLASSIFICATIONS:
Q. What is an
ultraviolet activator?
A.
It is a device that produces ultraviolet light intended to
polymerize
or take off resinous
dental pit and fissure sealants or restorative material by transmission of
light through a rod.
The scope of
this guidance includes devices that use light sources such as
quarts-tungsten-halogen lamps, light emitting diodes (LED's), and
xenon-plasma arcs, as well as laser energy sources.
RULE:
The FDA guidance does not include heat or light sources intended
exclusively for tooth bleaching procedures. These are classified under
2CFR 872.6475, Heat Source for Bleaching Teeth, product code EEG. These
devices are exempt from the premarket notification
procedures in subpart E of part 807 of this chapter, subject to the
limitations of 872.9. (This action being considered cosmetic)
Q. What is FDA's
authority over cosmetics?
A.
It is very important to understand that Congress passes the laws and
govern the
United States. To
put those laws into effect, Congress authorizes certain government
agencies, including the FDA to create and enforce the rules and
regulations, but only those as authorized under the law. A change in
FDA's statutory authority over cosmetics would require a full
congressional vote.
(It would take an
act of Congress)
Additional information
is also available at (FD&C Act, sec. 201(i)).
21 CFR 872.6070
Ultraviolet Activator for Polymerization |